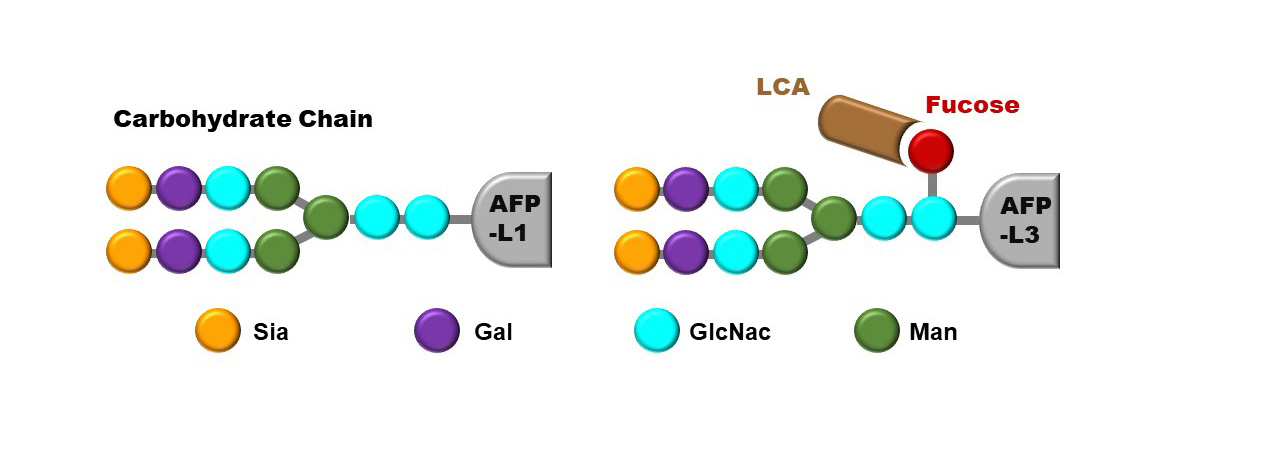
Lectin-reactive fraction of alpha-fetoprotein (AFP-L3) is a serum biomarker for hepatocellular carcinoma (HCC). Adding AFP-L3 to your current surveillance practice for HCC could increase your chances of detecting early HCC.
The μTASWako AFP-L3 Immunological Test System is an in vitro device that consists of reagents used with the μTASWako i30 Immunoanalyzer to quantitatively measure, by immunochemical techniques, AFP-L3% in human serum. The device is intended for in vitro diagnostic use as an aid in the risk assessment of patients with chronic liver disease for development of hepatocellular carcinoma (HCC) in conjunction with other laboratory findings, imaging studies and clinical assessment. Patients with elevated AFP-L3% values (>= 10%) have been shown to be associated with an increase in the risk of developing HCC within the next 21 months and should be more intensely evaluated for evidence of HCC according to the existing HCC practice guidelines in oncology.
Both AFP-L3 and DCP tests are available at major reference laboratories in the United States and are CMS reimbursed. Please contact Wako Diagnostics for more information on how to order the tests in Canada.
| CPT code | |
|---|---|
| AFP-L3 | 82107 |
| DCP | 83951 |